Research
We develop analytical and numerical methods to understand how living matter organizes itself — from individual molecules to tissues.
Cytoskeletal Networks
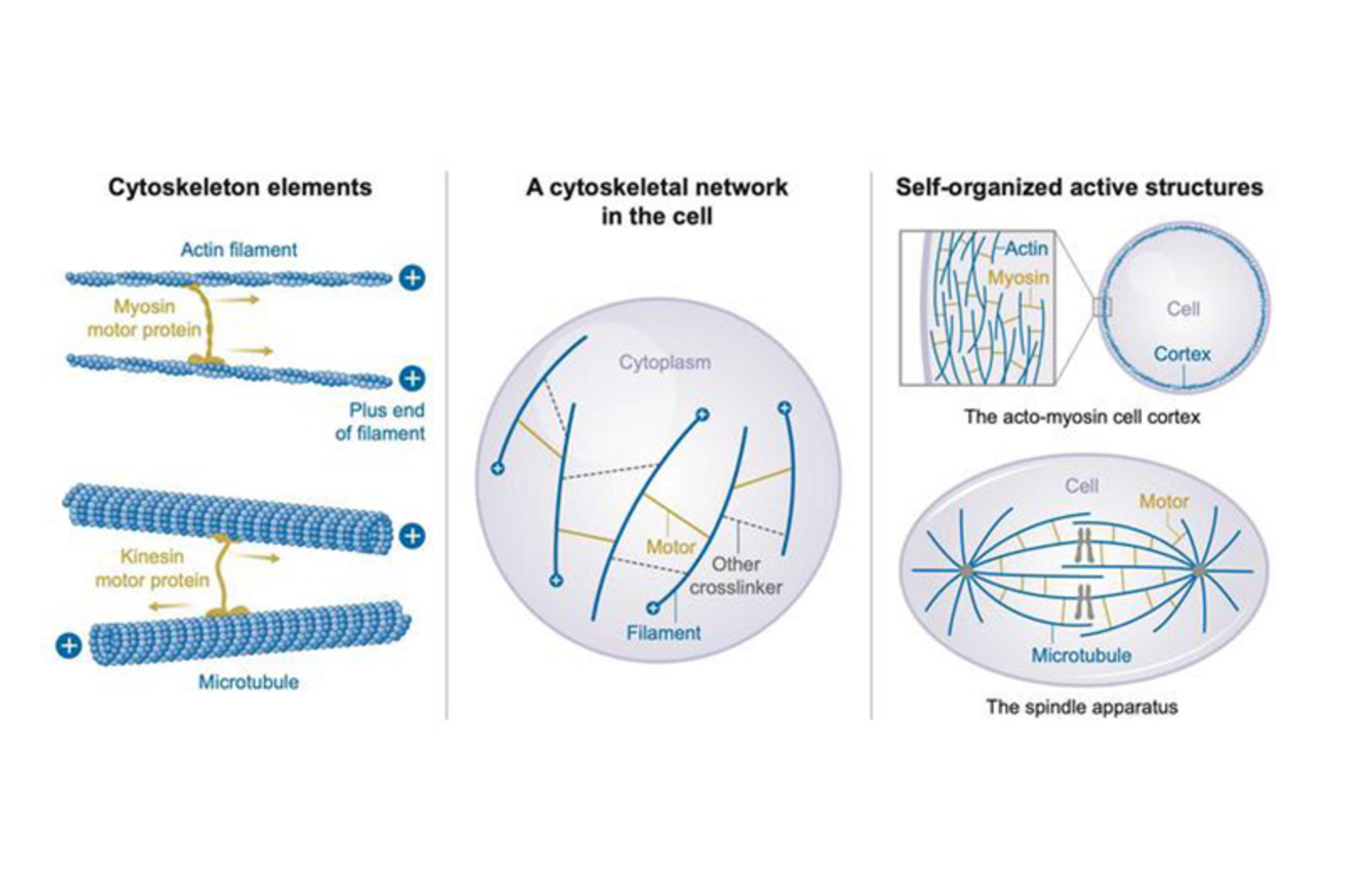
The cytoskeleton is the scaffold of the cell. Composed of filamentous proteins — actin, microtubules, and intermediate filaments — it enables cell shape changes, division, and motility. Crucially, it is an active material: molecular motors and associated proteins continuously consume energy to drive it out of thermodynamic equilibrium.
Phenomenological theories describe the cytoskeleton through effective material properties — viscosities, elasticities, and active stresses. But how these emerge from microscopic molecular interactions is often not well understood. Our group builds bridges between these scales.
Using analytical and numerical approaches, we develop frameworks for actively crosslinked filament networks. We derive how the number and dynamics of crosslinks shape the large-scale physical properties of these materials, providing quantitative predictions testable by experiment.
Key collaborators: Daniel J. Needleman (Harvard), Michael J. Shelley (Flatiron Institute).
Spindle Ultra-Structures

Every cell division requires the accurate segregation of duplicated chromosomes into two daughter cells. The mitotic spindle — a self-organized array of microtubules — is the machine responsible for this process. Errors in chromosome segregation lead to aneuploidy and are a hallmark of cancer.
Despite its importance, the physical principles governing spindle architecture are not fully understood. We integrate large-scale serial electron tomography with light microscopy data to reconstruct spindle ultra-structures at nanometer resolution, particularly in C. elegans.
From these structural datasets, we build physical models that explain how microtubule organization gives rise to the forces needed for chromosome capture, alignment, and segregation. We also study the physics of female meiosis, where the spindle must assemble without centrosomes.
Key collaborators: Stefanie Redemann (University of Virginia), Thomas Müller-Reichert (TU Dresden).
Centrosome Physics
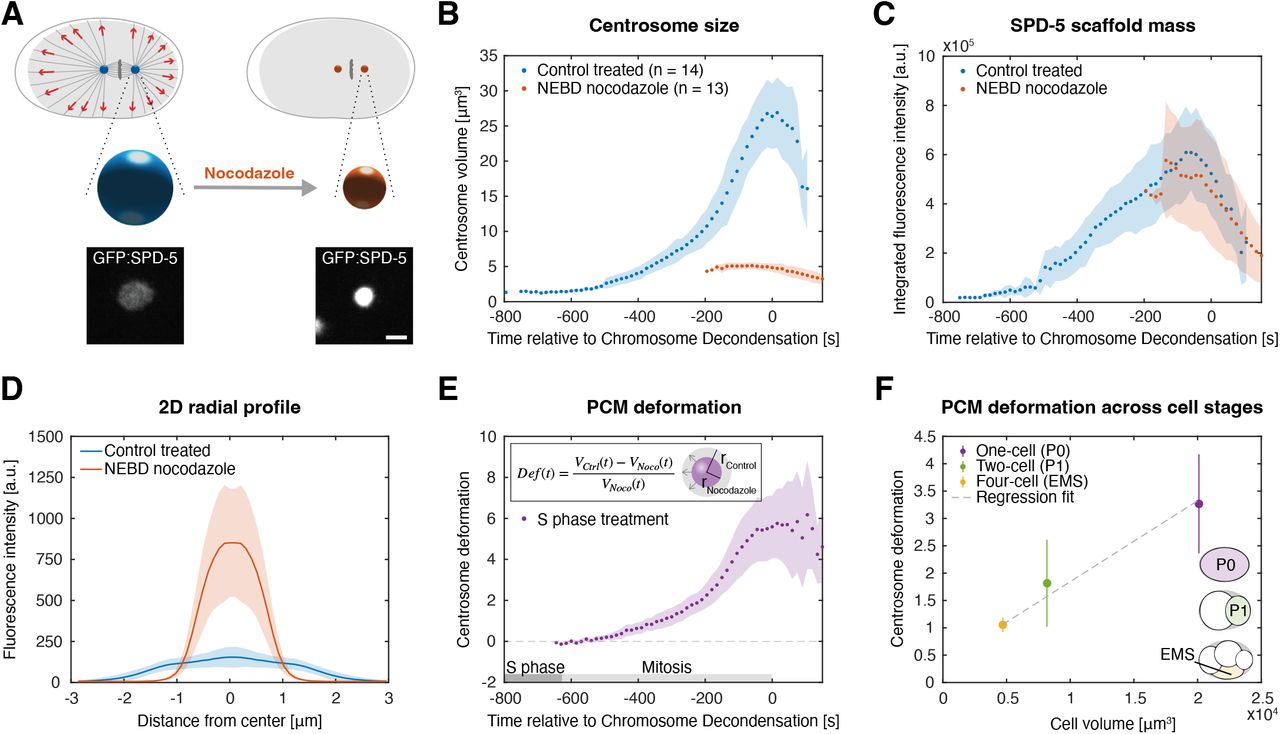
The centrosome is the primary microtubule-organizing center of animal cells. It consists of two centrioles surrounded by an amorphous cloud of proteins — the pericentriolar material (PCM) — which nucleates and anchors microtubules. Despite its central role in cell division, migration, and signaling, the physical principles that govern centrosome assembly and function remain poorly understood.
We investigate the mechanics of centrosome growth: how the PCM expands in a cell-cycle dependent manner, what forces this growth generates, and how those forces feed back onto the surrounding cytoskeleton. We are particularly interested in how growth-driven stresses contribute to centrosome positioning and to the organization of the mitotic spindle.
By combining continuum mechanical models with data from fluorescence microscopy, we aim to establish a quantitative physical picture of the centrosome as an active, self-organizing organelle.
Key team member: Emeline Gabrielle Laborie. Key collaborator: Alexander Dammerman.
Synchronization in Living Materials
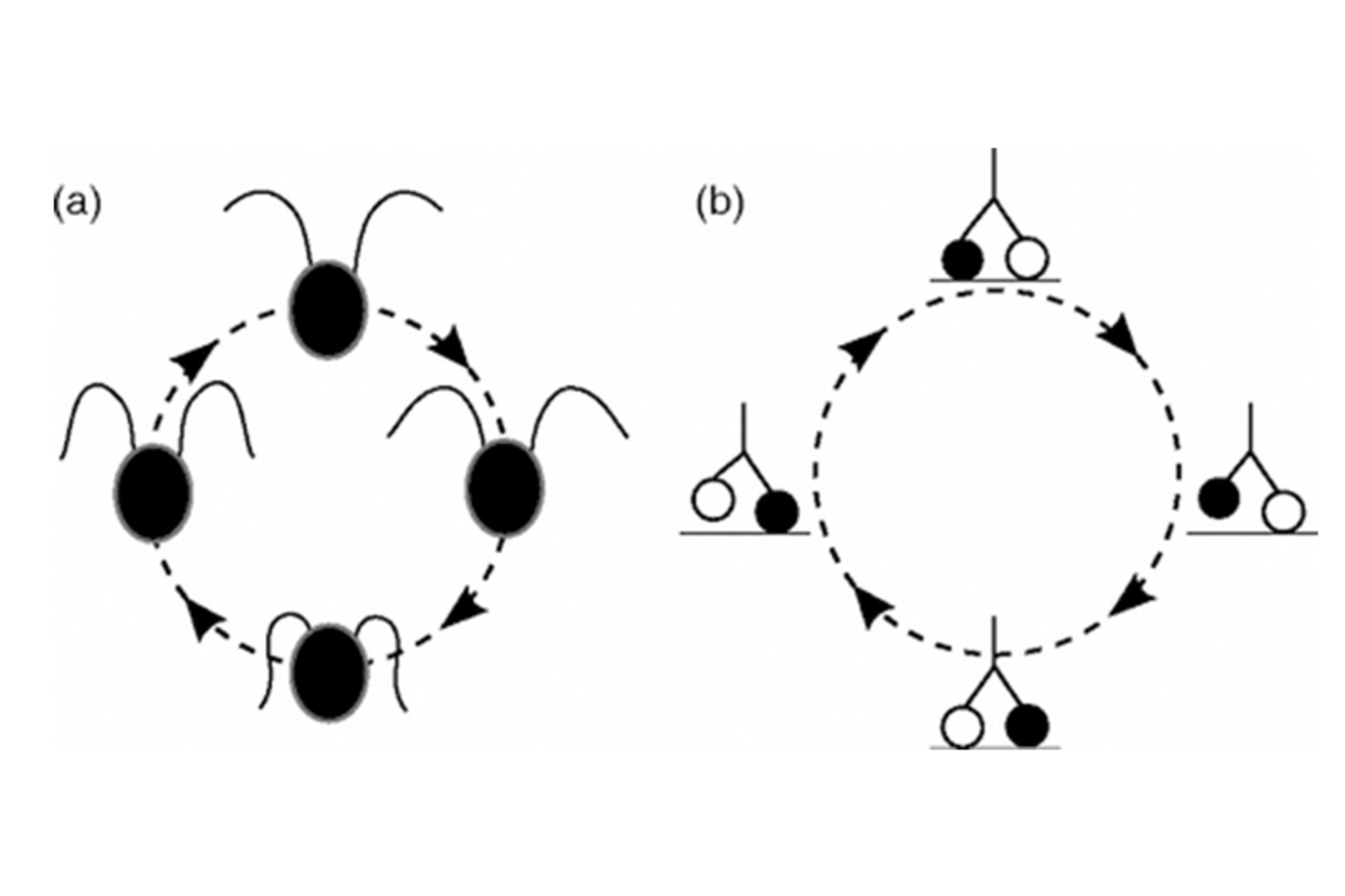
Molecular motors are protein machines that convert chemical energy — in the form of ATP hydrolysis — into mechanical work. They drive processes ranging from intracellular transport to the beating of cilia on epithelial surfaces.
A central question is how these nanoscale machines coordinate their activity across many length scales. Ciliated tissues lining the lungs, brain ventricles, and reproductive tracts must generate coherent metachronal waves — traveling patterns of coordinated ciliary beats — to efficiently pump fluid.
We build multiscale biophysical models that connect the mechanics of individual cilia to the collective dynamics of ciliary arrays, explaining how quantized wave patterns emerge from physical principles. We also study synchronization in bulk active fluids and develop continuum theories for phase-synchronized active materials.
Key collaborators: Brato Chakrabarti, Michael J. Shelley (Flatiron Institute), Sriram Ramaswamy.